
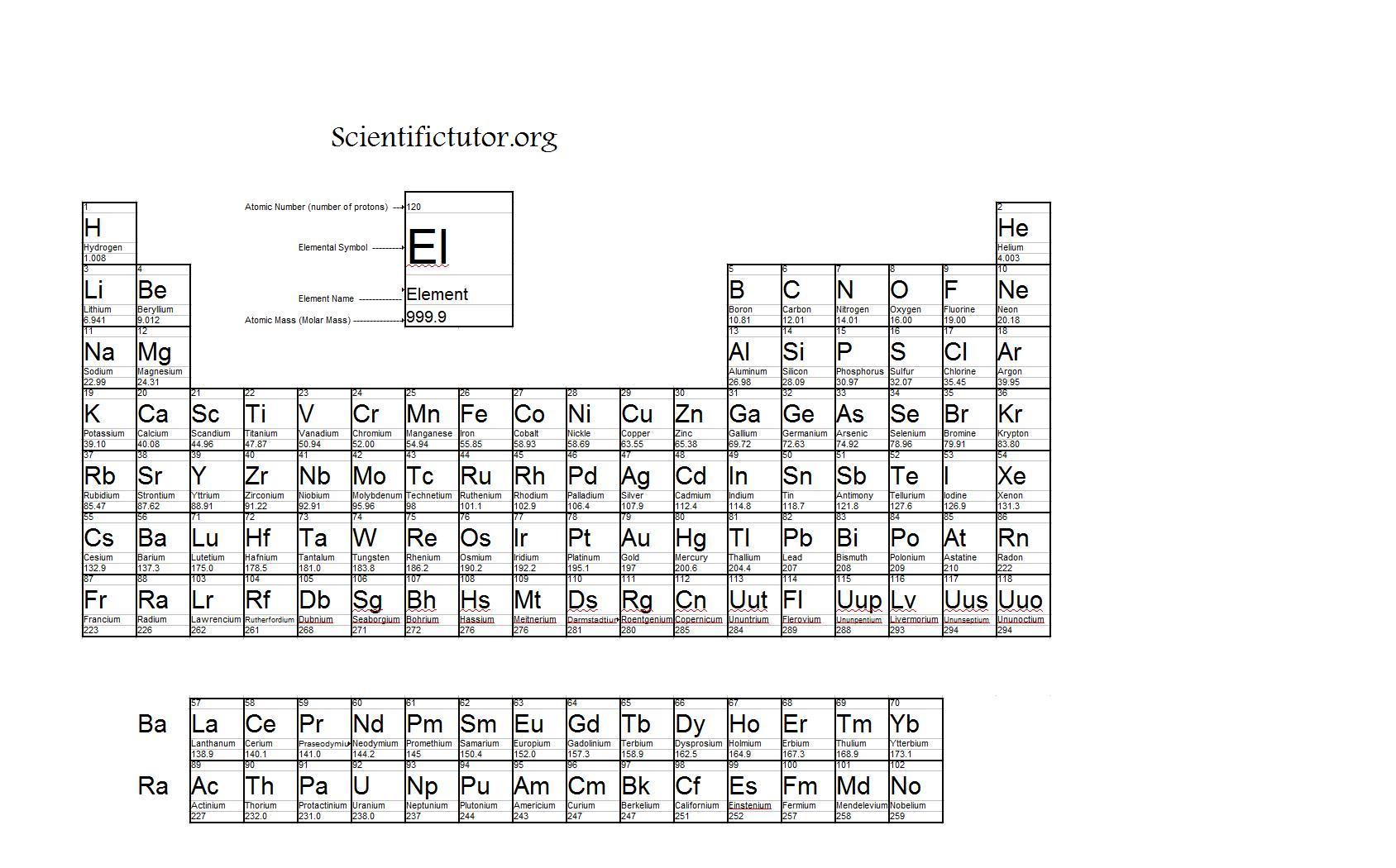
While 2 atoms with the same number of protons and differing numbers of neutrons are both the same element, they are different isotopes of that element. The number of neutrons can vary among atoms of a certain element. Though, especially among elements at the top of the periodic table, it may seem that an atoms' atomic mass is about twice its atomic number, atomic mass isn't ever calculated by doubling an element's atomic number.įind the number of neutrons in the nucleus. Note that an element's atomic number doesn't have any direct bearing on its relative atomic mass as listed on the periodic table.We can also see on the periodic table that the square for carbon (C) has a "6" at the top, signifying that carbon's atomic number is 6. Carbon always has 6 protons, so we know its atomic number is 6. Let's say that we're working with the carbon atom.This number will always be a positive whole number. You can find the atomic number of any element on the periodic table - in nearly all standard periodic tables: it's the number above an element's 1 or 2-letter chemical symbol. Sodium has an atomic number of 11 because its nucleus has 11 protons, while oxygen has an atomic number of 8 because its nucleus has 8 protons. 
X Research source For example, all hydrogen atoms, and only hydrogen atoms, have 1 proton. The atomic number is the number of protons in an element, and never varies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
